Abstract
In chiral chromatography, the technology of detecting chiral molecules using optical devices has lagged behind until now. The stability and sensitivity of this type of instrument is an important issue in chiral chromatography. However, with the availability of the single wavelength LED, increased sensitivity and increased economics with simplified instrument design was possible. In addition, a newly designed glass coated steel flow cell has produced a superior, third generation optical rotation detector. Aided with the Faraday compensation technique, this instrument gives high optical rotation power with excellent signal to noise ratio.
This instrument is an excellent supplemental detector to standard UV detection.
For chiral separations, it permits the identification of elution order which is important when quantifying to establish minimum levels of detection. It allows for the identification of enantiomeric pairs when dealing with multiple chiral centers. During the method development process, this detector detects marginal selectivity when UV detection fails. The important features of this instrument will be described. Also, examples of absolute sensitivity, identification of enantiomer pairs and reversed elution order will be included.
Polarimetry and Chiral Detection
- Identifies the chirality of a molecule with a positive or negative sign of rotation.
- Detects only optically active compounds.
- Can detect optically active compounds with no or low UV absorbance.
- Works with gradients.
- Simple determination of enantiomeric excess (ee) and optical purity.
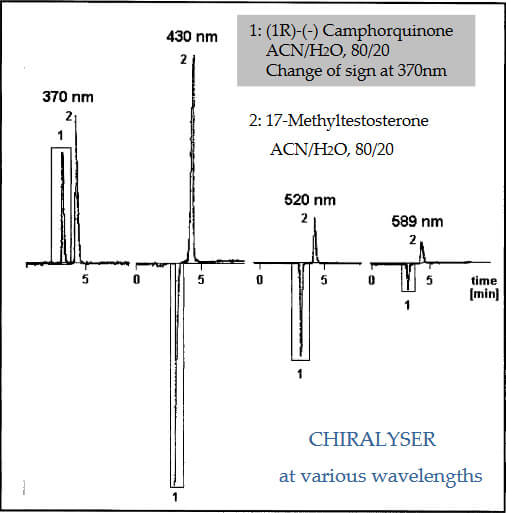
Optical Rotation as a Function of Wavelength
At 430nm from a high power blue LED, the maximum optical rotation dispersion is obtained with minimum absorption effects.
Although intensity is lower than laser sources the distribution is constant with no interference effects (low noise) commonly seen with laser sources.
Faraday Effect
The Faraday effect describes the rotation of the plane of polarized light produced when plane polarized light is passed through a substance in a magnetic field, the light traveling in a direction parallel to the direction of propagation. For a given substance, the angle of rotation is proportional to the length of the column which the light traverses and the strength of the magnetic field.
Since a compensating current can be made proportional to the optical rotation of the sample, the analysis is in real time simpler and substantially faster than the mechanical manipulation of previous devices.
The Faraday modulation measures the rotation angle independent of the light source intensity or absorption of the sample. This fast electrical compensation is proportional in real time to the amount of optically active substance inside the cell which is easily transformed to a calibrated readout.
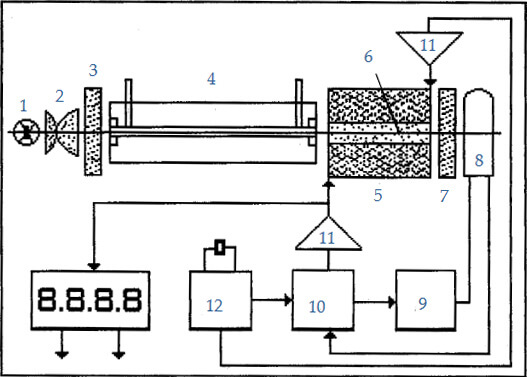
1. LED, 430nm
2. Lenses
3. Polarizer
4. Cell
5. Coil
6. Rod
7. Analyser
8. Photomultiplier
9. HV‐Generator
10. Phase Sensitive Integrator
11. Amplifier
12. Time Base
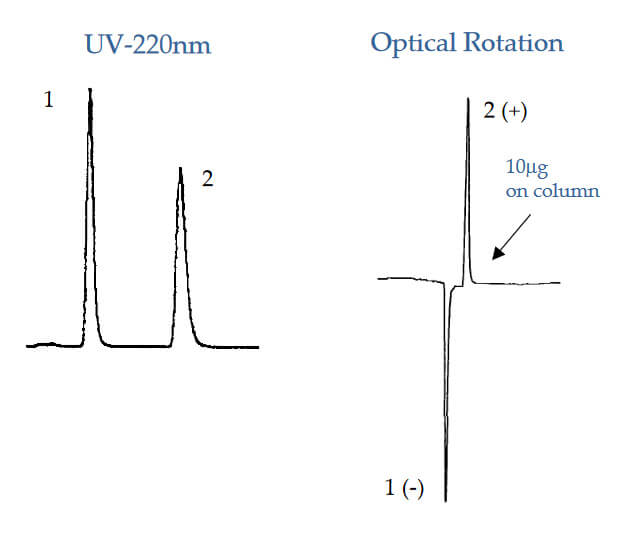
UV vs Optical Rotation
Analyte: 5‐Methyl‐5‐phenylhydantoin
Column: CHIROBIOTIC T, 250×4.6mm
Mobile Phase: 100% Methanol
Flow Rate: 1.0 mL/min.
Identifying Reversal of Elution Order
Analyte: Propranolol
Column: CHIROBIOTIC T and CHIROBIOTIC TAG, 250×4.6mm
Mobile Phase: 100/0.1%wt.:MeOH/ATFA
Flow Rate: 1.0 mL/min.
CHIROBIOTIC T
CHIROBIOTIC TAG
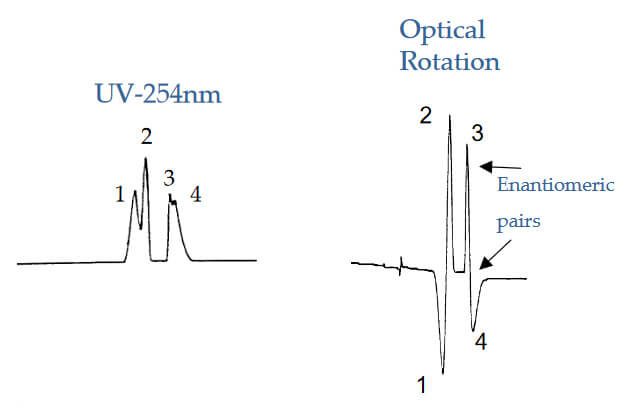
Identification of Enantiomeric Pairs
Analyte: Labetalol
Column: CHIROBIOTIC V, 250×4.6mm
Mobile Phase: 100/0.1%wt.:MeOH/AFTA
Flow Rate:1.0 mL/min.
Peak 1 (+);Peak 3 (‐) Enantiomers
Peak 2 (+);Peak 4 (‐) Enantiomers
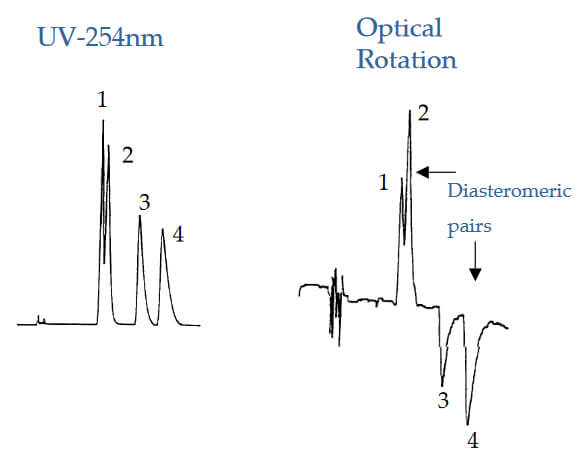
Identification of Enantiomeric Pairs
Analyte: Unknown Sample (2 chiral centers)
Column: CHIROBIOTIC V
Mobile Phase: 100/0.15/0.05; MeOH/HOAc/TEA
Flow Rate: 1.0 mL/min.
Peak 1(‐); Peak 2 (+) Enantiomers
Peak 3 (+); Peak 4 (‐) Enantiomers
Determination cis/trans Enantiomers
Column: CYCLOBOND I 2000 RSP, 250×4.6mm
Mobile Phase: 40/60: ACN/5mM NH 4OAc, pH 3.8
Flow Rate: 0.8 mL/min.
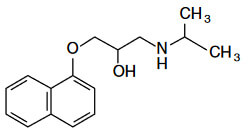
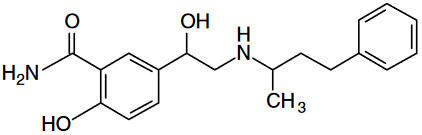
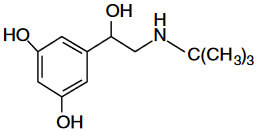
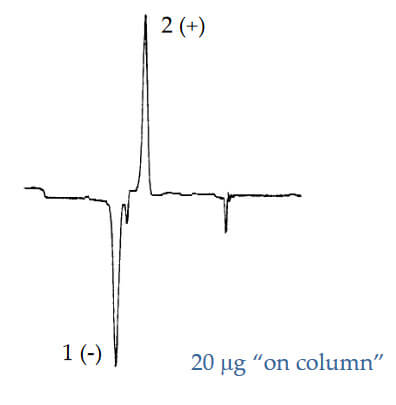
Optical Rotation of Amino Alcohol
Analyte: Terbutaline
Column: CHIROBIOTIC T, 250×4.6mm
Mobile Phase: 100/0.1%w: MeOH/ATFA
Flow Rate: 1.0 mL/min.
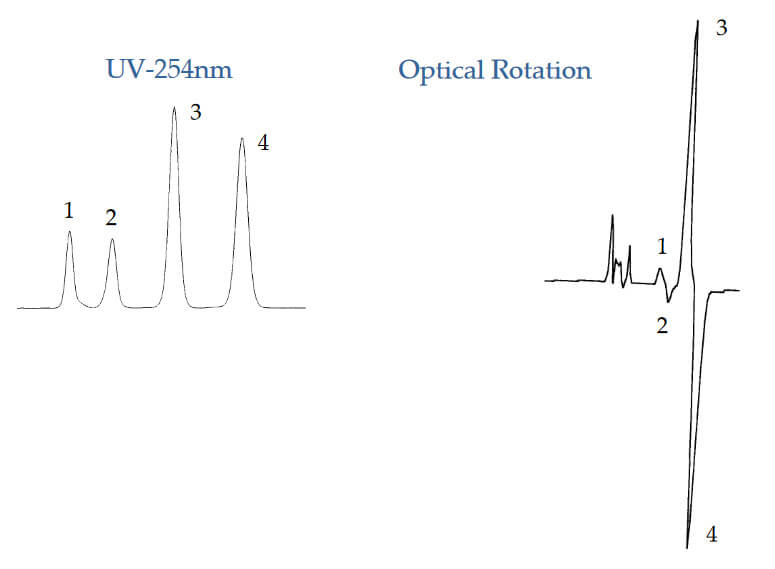
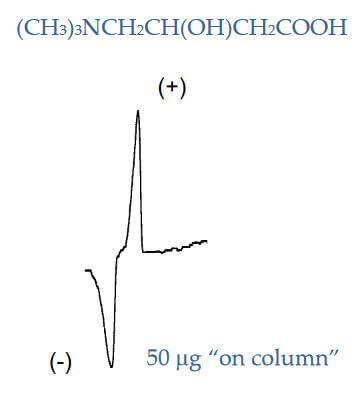
Determining Rotation of Analyte with No UV
Analyte: Carnitine
Column: CHIROBIOTIC TAG, 250×4.6mm
Mobile Phase: 85/15: MeOH/20mM NH 4OAC, pH 6.0
Flow Rate: 1.0 mL/min.
Polarimetry, unlike circular dichroism does not require that the analyte have a detectable UV wavelength. This makes it a very useful tool for detecting non‐aromatic amino acids and other similar substances.
Conclusions
A stable, sensitive, economical and simple polarimetry device has been described with a 430nm ‘blue’ LED light source for optimal rotation determinations.
This device is a useful aide to chiral chromatography work by providing:
- The sign of rotation of chiral molecules with or without UV chromaphores.
- Information on any potential for a reversal of elution order.
- Information on the presence of enantiomeric pairs or diasteriomers.
- Optical purity/enantiomeric excess.
Thomas E. Beesley and J.T. Lee
Advanced Separation Technologies Inc. (Astec)
37 Leslie Court, P. O. Box 297
Whippany, New Jersey 07981 USA
Website: www.astecusa.com; Email: info@astecusa.com
Wolfgang Zelenka
IBZ Messtechnik GmbH
Mozartstrasse 14‐16,Lower Saxony, Hannover D‐30173,Germany
Poster # 21700‐400P